Companies who produce or handle medicines/drugs often need to do qc measurements on products they produce, process or handle for various reasons. This can be either incoming qc, which is often anti counterfeit. Or it can be outgoing QC. Most methods that are used which take time and need sample preparation. Raman can offer a fast analysis alternative with zero or minimal sample preparation.
Why Wasatch
Often companies initially investigate benchtop Raman systems. The Wasatch Raman spectrometers are able to compete in performance for this application. Price wise a Wasatch system will be 3-6 times lower compared to a benchtop Raman spectrometer. So when working on an application that needs a Raman Fingerprint of Medicines, Wasatch Photonics is your go-to partner.
Choice of wavelength
There is a diverse variation of pills, liquids, suspensions, etc. Many white pills contain Microcrystalline cellulose which exhibits some fluorescence, often the Raman signal is stronger than the fluorescence. Some pills have coatings, which can be a wax or a sugar, with these pills minor sample preparation ‘might’ be needed to remove the coating. Talk to your prospect about the samples that need to be measures and gather as much information as you can.
There is not one wavelength that can measure all medicines. In publications 785 nm is most often used, we believe this has more to do with availability of equipment. It is likely that overall 830 nm Raman systems will show least fluorescence and give the optimal performance.
Samples
Raman Fingerprint of Medicines can be measured in either solid or in liquid form. Measurements can be done either directly on the sample, or trough the wall of the vial that the sample is in.
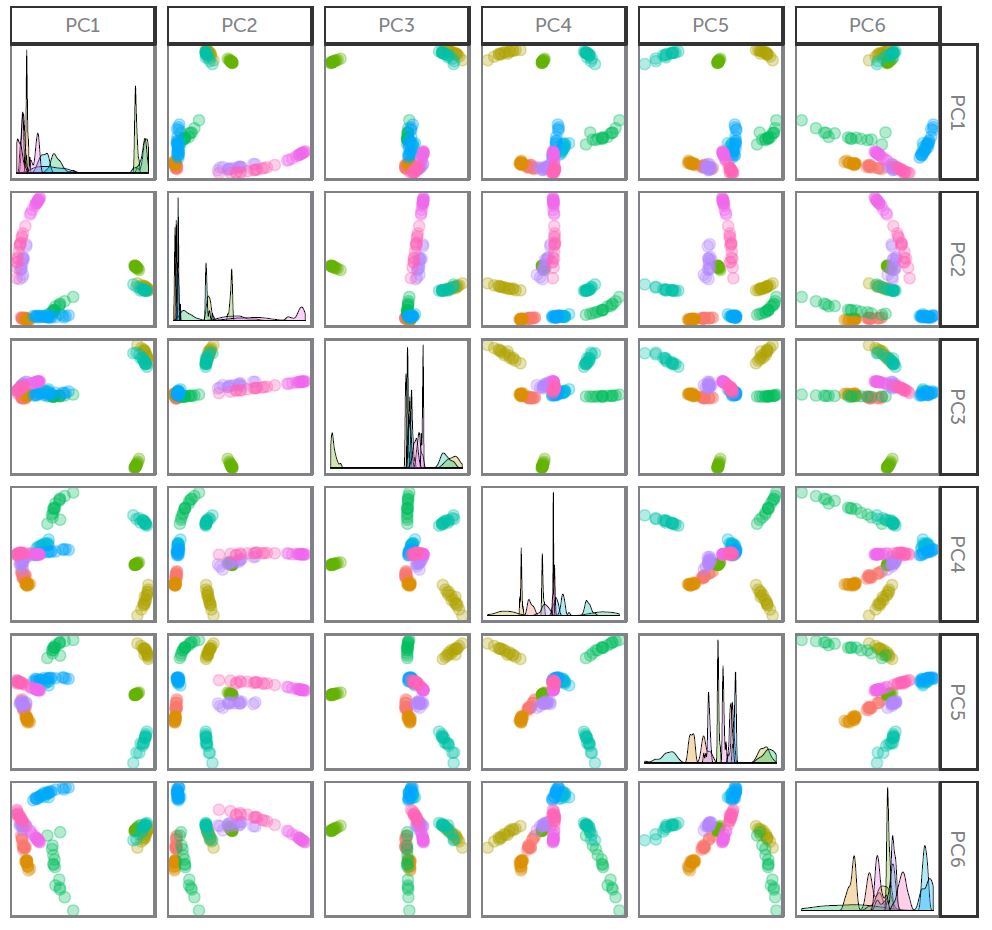
PCA Analysis results using a Wasatch Raman spectrometer measuring medicine samples
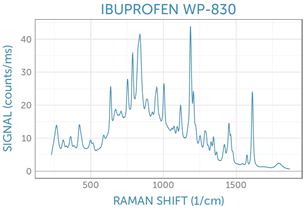
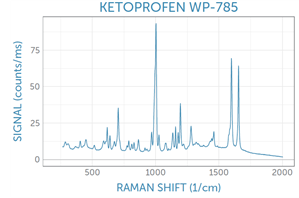
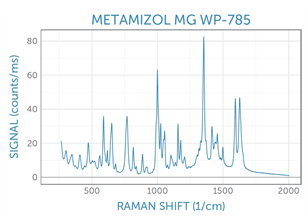
Example spectra
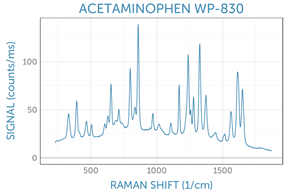
Thank you for reading.
#Raman #spectrometer #pharma #medicine #wasatch

